
Mindfulness-based Cognitive Therapy to Improve Sleep Quality in Older Adults with Insomnia
[Terapia cognitiva basada en mindfulness para mejorar la calidad del sueño en adultos mayores con insomnio]
Manuel Camino1, Encarnacion Satorres1, Iraida Delhom2, Elena Real1, Mireia Abella1, and Juan C. Meléndez1
1University of Valencia, Spain; 2Universidad Internacional de Valencia, Spain
https://doi.org/10.5093/pi2022a12
Received 15 February 2022, Accepted 6 June 2022
Abstract
There is a high prevalence of insomnia in older adults, which has negative consequences for their well-being and quality of life. The recommendation for first-line treatments is to administer non-pharmacological interventions. The objective of this research was to verify the effectiveness of Mindfulness-Based Cognitive Therapy in older adults with subclinical and moderate insomnia, studying its impact on sleep quality. One hundred and six older adults participated and were assigned to the subclinical insomnia (n = 50) or moderate insomnia (n = 56) groups; subsequently, they were randomly assigned to the control and intervention groups. Subjects were evaluated at two times with the Insomnia Severity Index and the Pittsburgh Sleep Quality Index. Significant results were obtained on both scales, with a reduction in insomnia symptoms in the subclinical and moderate intervention groups. The administration of a treatment that combines mindfulness and cognitive therapy is effective for treating insomnia in older adults.
Resumen
Existe una alta prevalencia de insomnio en los adultos mayores, lo que tiene consecuencias negativas para su bienestar y calidad de vida. La recomendación para los tratamientos de primera línea es administrar intervenciones no farmacológicas. El objetivo de esta investigación fue verificar la efectividad de la terapia cognitiva basada en mindfulness en adultos mayores con insomnio subclínico y moderado, estudiando su impacto en la calidad del sueño. Participaron 106 adultos mayores que fueron asignados a los grupos de insomnio subclínico (n = 50) o de insomnio moderado (n = 56); posteriormente, fueron asignados aleatoriamente a los grupos de control y de intervención. Los sujetos fueron evaluados antes y después de la intervención con el índice de gravedad del insomnio y el índice de calidad del sueño de Pittsburgh. Se obtuvieron resultados significativos en ambas escalas, con una reducción de los síntomas de insomnio en los grupos de intervención subclínica y moderada. La administración de un tratamiento que combina mindfulness y terapia cognitiva es eficaz para tratar el insomnio en adultos mayores.
Keywords
Subclinical insomnia, Moderate insomnia, Aging, Sleep qualityPalabras clave
Insomnio subclĂnico, Insomnio moderado, Envejecimiento, Calidad del sueñoCite this article as: Camino, M., Satorres, E., Delhom, I., Real, E., Abella, M., & Meléndez, J. C. (2022). Mindfulness-based Cognitive Therapy to Improve Sleep Quality in Older Adults with Insomnia. Psychosocial Intervention, 31(3), 159 - 167. https://doi.org/10.5093/pi2022a12
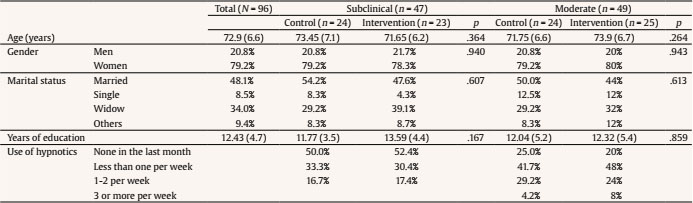
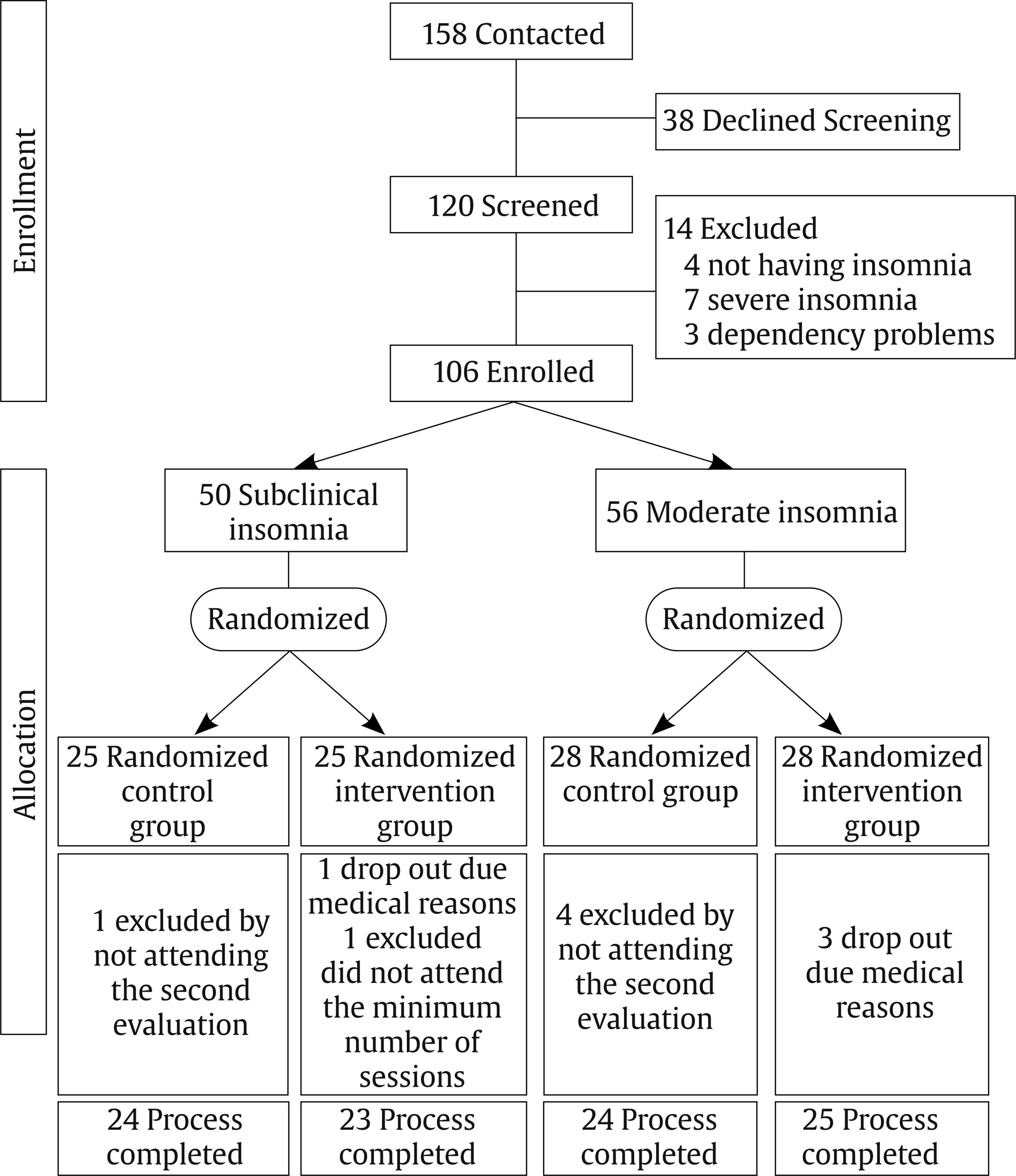
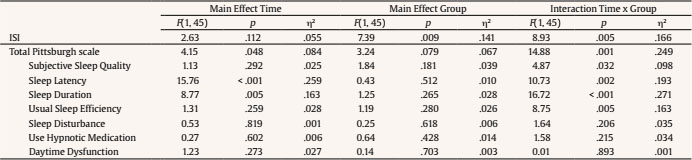
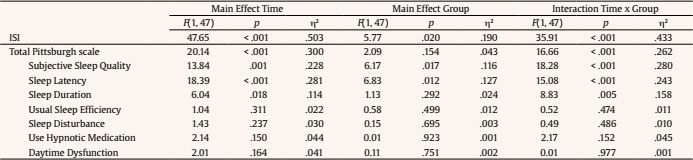
melendez@uv.es Correspondence: melendez@uv.es (J. C. Melendez).The fifth edition of the Diagnostic and Statistical Manual for Mental Disorders (DSM-5) (American Psychiatric Association [APA, 2013]) broadly defines insomnia as a predominant dissatisfaction with the quantity or quality of sleep under favorable sleeping conditions, associated with one (or more) of the following symptoms: difficulty initiating sleep, difficulty maintaining sleep, and early-morning awakening with an inability to return to sleep. This definition emphasizes that a sleep disturbance causes clinically significant distress or functional impairment, despite having an adequate opportunity to sleep. One of the populations most likely to suffer from insomnia is older adults. Epidemiological reports of insomnia vary somewhat depending on the diagnostic criteria used, but the overall prevalence of insomnia symptoms ranges from 30% to 48% in older adults, whereas the prevalence of insomnia disorder ranges from 12% to 20% in the general population (Patel et al., 2018). Brewster et al. (2018) showed that the incidence of insomnia increases with age, and that sleep efficiency declines after the age of 60. The meta-analysis carried out by Ohayon et al. (2004) obtained empirical evidence supporting the relationship between age and sleep quality, demonstrating that sleep parameters change significantly with age across the adult lifespan and show deterioration. Total sleep time, sleep efficiency, and deep sleep (slow-wave sleep) decrease with aging, and the number of nocturnal awakenings and the time spent awake during the night increase with age (Li et al., 2018). However, sleep latency and the ability to fall back to sleep after nocturnal awakenings show minimal modifications after the age of 60 (Patel et al., 2018). Insomnia in older adults has important consequences that affect different areas of health. Berkley et al. (2020) report the negative impact on quality of life and daily functioning, affecting cognitive, physical, and emotional health. Insomnia is a consistent predictor of poor physical and mental health (Biddle et al., 2019; Fernández-Mendoza & Vgontzas, 2013). It has also been proposed as a predictor of cognitive impairment. Sexton et al. (2020) obtained mixed evidence for impaired performance on objective cognitive tests and reduced grey matter volumes. The meta-analysis by Hertenstein et al. (2019) found that insomnia is a significant predictor of depression, anxiety, and alcohol abuse. The sleep loss resulting from sleep disturbances can lead to daytime sleepiness, chronic fatigue, and increased risk of falls and accidents (Foley et al., 1995), as well as an increased risk of frailty in older adults (Pourmotabbed et al., 2020). Finally, the results found by Dragioti et al. (2018) agreed with previous findings from longitudinal and cross-sectional studies showing that insomnia severity is a potential indicator of low general well-being and quality of life. Treatments for older adults with insomnia may include (a) psychological therapies, (b) pharmacotherapy, or (c) a combination of the two, depending on the severity of the symptoms. The European guidelines state that there are high levels of evidence for the short-term use of benzodiazepines and hypnotic benzodiazepine receptor agonists in insomnia disorder, but long-term treatment is specifically advised against (Riemann et al., 2017). Benzodiazepines are able to reduce sleep latency and increase total sleep time in only two weeks (Gunja, 2013), but at the expense of reducing REM sleep and deep sleep phases, fundamental variables in restful sleep. Furthermore, the association between long-term benzodiazepine use and risk of dementia has received significant attention. A more recent systematic review of observational studies (cohort and case-control studies) found that long-term users of benzodiazepines have a 1.5- to 2-fold increased risk of developing dementia, compared with non-users (Billioti de Gage et al., 2015; Zhong et al., 2015). The impact of this problem on the quality of life of older adults should not be underestimated. Considering the possible negative impact of drug use on quality of life, according to Kamel and Gammack (2006), the solution lies in the administration of mainly pharmacology-free interventions, resorting to pharmacology only when necessary as a second line of treatment. Riemann et al. (2017) reported that, due to the strong evidence supporting cognitive behavioral therapy for insomnia (CBT-I), it is considered a first-line treatment for chronic insomnia in adults of any age (strong recommendation, high-quality evidence) that is safe and effective and has stable long-term effects (Frase et al., 2018). CBT-I targets factors that may maintain insomnia over time, such as dysregulation of the sleep drive, sleep-interfering behaviors and cognitions, attempts to control the sleep process, and sleep-related anxiety (Baglioni et al., 2020). It is a multi-component treatment for insomnia that typically consists of psychoeducation/sleep hygiene, relaxation training, stimulus control therapy, sleep restriction therapy, and cognitive therapy (Dzierzewski et al., 2018). CBT-I has demonstrated efficacy, with behavioral strategies aimed primarily at restoring sleep regulation and cognitive components aimed at restructuring maladaptive sleep-related cognitions (Ong & Smith, 2017). However, patients who maintained pre-sleep arousal seemed to have a greater probability of reoccurring insomnia, requiring the administration of other alternative therapies that, in addition to the objectives of the CBT-I, impact the dysregulation of arousal (Ong et al., 2009). Belanger et al. (2016) suggest that 20-30% of patients do not achieve clinically significant improvement following CBT-I; furthermore, only about 40% of CBT-I recipients achieve clinical remission (Morin & Benca, 2012). Mindfulness-based therapies (MBT) offer one of these alternative approaches. Mindfulness interventions for insomnia were initially based on the original Mindfulness-Based Stress Reduction (MBSR) program developed by Kabat-Zinn (2003). Significant improvements were found from baseline to post-treatment on measures such as the Insomnia Severity Index or the Pittsburgh Sleep Quality Index (Gross et al., 2011; Zhang at al., 2015). Subsequently, Segal et al. (2002) developed Mindfulness-Based Cognitive Therapy (MBCT) with the intention of using mindfulness principles to disrupt negative ruminative thought patterns that contribute to depression relapse. The MBCT is a program that arose from the MBSR, adding cognitive therapy components to reduce depression. CBT is relevant for the treatment of insomnia, helping to improve control of thoughts and reduce rumination that hinders sleep. Therefore, it makes sense to address the use of MBCT for the treatment of insomnia in a population without depression to increase sleep time and decrease sleep latency (Foulk et al., 2014; Heidenreich et al., 2006). In addition, there are other reasons for using this model of intervention strategies for insomnia (Shallcross & Visvanathan, 2016): a) MBCT trains experiential awareness, attentional control, and acceptance, variables that are associated with poor sleep (Ong et al., 2012; Woods et al., 2009); b) insomnia has a comorbid relationship with depression, and MBCT has extensively shown its effectiveness in preventing depression and reducing depressive symptoms (Shallcross et al., 2015; Williams et al., 2014); and c) skills that are trained in MBCT have implications for well-being that are maintained over time and may be related to improving insomnia (Mathew et al., 2010). Due to the lack of research on psychological therapies in older adults with insomnia, the main goal of this research was to find out whether the administration of a MBCT program could have relevant effects on sleep quality. Showing the efficacy of this intervention would foment its use to prevent and treat the complications in this population. For this purpose, two specific groups of insomnia were selected: subclinical and moderate. Some authors (Low et al., 2020; Morin et al., 2006) suggest that many individuals with insomnia do not seek professional help and instead tend to use self-help strategies. It is generally those subjects with more severe and chronic insomnia who make medical consultations (Morin et al., 2006). This is particularly important given the large proportion of individuals in the community with subclinical insomnia. Moreover, there is an increased likelihood that subclinical insomnia symptoms will progress to clinical levels if left untreated (Morin & Jarrin, 2013) and that subjects with moderate insomnia may progress to severe insomnia. Given these possible increases in insomnia rates, we decided to investigate the efficacy of mindfulness-based interventions in subclinical and moderate insomnia. The main hypothesis was that intervention participants in the subclinical insomnia and moderate insomnia groups would show a decrease in the insomnia dimension and an increase in sleep quality compared to control groups belonging to the same insomnia category. Furthermore, we hypothesized that the intervention would have more significant effects in the moderate group than in the subclinical group. Participants Of the 158 people contacted for the study, 38 declined to be screened. The inclusion criteria were: age over 60 years old, able to read, with no visual problems that would make it difficult to see images, and scores between 8 and 21 on the Insomnia Severity Index (ISI; Morin, 1993), which categorizes subjects’ insomnia from subclinical to moderate. The exclusion criteria were not being institutionalized or presenting high dependency, and no presence of significant asymptomatic neurovascular disease, history of previous symptomatic stroke, a medical condition that significantly affects the brain, severe psychiatric symptoms, attended at least 80% of the sessions, or scores lower than 6 on the Pittsburgh Sleep Quality Questionnaire (PSQI; Buysse et al., 1989), which indicates “good sleepers”. In addition, subjects who were categorized, based on their score on the ISI scale, as not having insomnia or as having severe insomnia were excluded because those with severe insomnia symptoms would require specific interventions to alleviate the severity of their symptoms. Finally, as a requirement to participate, they had to fully complete the evaluation protocol prior to the intervention. Fourteen were excluded during screening (application of inclusion/exclusion criteria): 4 for not having insomnia, 7 for having severe insomnia, and 3 due to dependency problems. Finally, 106 subjects participated in the study and were assigned to the subclinical insomnia group (n = 50) and to the moderate insomnia group (n = 56) based on Insomnia Severity Index criteria. All participants were informed about the study objectives and signed the respective informed consent. Once subjects were included in the insomnia groups (subclinical or moderate), they were randomly assigned following simple randomization procedures to the control and intervention groups (see Figure 1). The study follows the principles of the Declaration of Helsinki. From the initial subclinical group, one control dropped out of the process by not attending the second evaluation and two dropped out of the intervention group: one due to medical reasons during the intervention and the other because s/he did not attend the minimum number of established sessions. From the initial moderate group, four controls dropped out by not attending the second evaluation, and three participants in the intervention group dropped out due to medical reasons during the process. Thus, the final subclinical insomnia group consisted of 47 participants (24 control group and 23 intervention group) and the final moderate insomnia group consisted of 49 participants (24 control group and 25 intervention group). Table 1 presents the sociodemographic data of the groups as well as the contrast tests of the groups to demonstrate their equality on these characteristics (see Table 1). Instruments The Insomnia Severity Index (ISI; Morin, 1993) is a brief, self-report instrument to assess the patient’s perception of insomnia based on the criteria of the Diagnostic and Statistical Manual of Mental Disorders DSM-IV and the International Classification of Sleep Disorders (American Sleep Disorders Association [ASDA, 1997]). The ISI focuses on the subjective symptoms and consequences of insomnia, as well as the degree of worry or distress caused by these difficulties. This measure assesses night-time sleep difficulties and sleep dissatisfaction, but also the impact of the disorder on daytime functioning (e.g., daytime fatigue or ability to function at work). The scale was validated in Spanish for older adults (Fernández-Mendoza et al., 2012). The total score on the questionnaire is divided as follows: 0-7, no significant insomnia; 8-14, subclinical insomnia; 15-21, moderate insomnia; and 22-28, severe insomnia. The Pittsburgh Sleep Quality Index (PSQI; Buysse et al., 1989) is the most widely used sleep health assessment tool in both clinical and non-clinical populations, and it assesses qualitative and quantitative aspects of sleep quality in the month prior to its application. Self-reported items are used for the quantitative assessment of patient-perceived sleep quality and to evaluate specific features: subjective quality, latency (understood as the time the patient thinks it takes him/her to fall asleep), duration, usual efficiency (assesses the percentage of time the patient believes he/she is asleep over the total time he/she remains lying down), disturbances (alterations such as pain, cold, cough, etc.), the use of hypnotics, and daytime dysfunction (presented as the ease of falling asleep while doing some activity or as increased daytime fatigue). Each of these components is assigned a discrete score that can range from 0 to 3; a score of 0 indicates that there are no problems in this regard, whereas a score of 3 indicates serious problems. The PSQI was validated in Spanish with good psychometric results (Hita-Contreras et al., 2014). Procedure Initially, telephone contact was established with different care centers for older adults in the city of Valencia (CEAM). The possibility of administering an intervention based on mindfulness to improve sleep quality was proposed to them. With the centers that showed interest in the proposal, mechanisms were established to initiate collaboration. After this initial contact, an individual meeting was held with each of the directors of the centers, as well as with the social workers, where the objectives of the study were explained to them. Posters were displayed, and brochures explaining the study objectives and enrollment procedure were left at the participating senior centers so that their users could read them. Interested persons went to the center’s Care Office and filled out a brief data form which they placed in an envelope and left for later telephone contact (158 subjects). During the telephone contact, more information was provided about the study and the intervention to be applied; 38 refused to participate; those who refused screening were not asked for their reasons. People who were interested were scheduled for first session. This first session was designed to explain how the intervention was going to be structured, resolve participants’ doubts, evaluate and screen participants to verify compliance with the inclusion criteria, sign the informed consent, and create a comfortable stay. Finally, 106 people were enrolled in the study, and the allocation phase began with the identification of the subjects as having subclinical or moderate insomnia based on Insomnia Severity Index criteria. Subsequently, for each category of insomnia, a simple randomization was performed, and subjects were assigned to the intervention and control groups. The controls carried out an eight-week cine-forum activity (coinciding with the weeks of the intervention) about active ageing; in addition, they were asked to be included on a waiting list for the subsequent administration of the intervention. After the activity was over, they were also called to a group session to carry out the post-intervention evaluation. The intervention group attended eight intervention sessions following the MBCT guidelines, plus a final evaluation session. The intervention administered was the same for the subclinical and moderate insomnia groups. The idea for administering the same intervention was to test whether a specific intervention really had an effect on these groups of insomniacs, albeit with significantly different symptoms. There is scientific evidence demonstrating the efficacy of mindfulness-based therapies in the treatment of sleep problems (Rusch et al., 2019). In addition, MBCT programs generally include a part with cognitive therapy (Williams & Kuyken, 2012), which could help to reduce the ruminative thoughts that occur in sleep disorders. Therefore, the program administered was based on these two types of interventions combined. The intervention was administered based on an active methodology with structured sessions. For its administration, a space conducive to reflection, psychoeducation, and mindfulness was promoted. The philosophy of the intervention highlights that the participants are the main agents of action, given that competencies are acquired through training. A psychologist was the professional entrusted with carrying out the mindfulness training with the participants. Furthermore, he/she had experience with MBCT in clinical contexts and as a mindfulness instructor in several mindfulness retreats. Each group had between 9 and 16 participants. Each working session lasted an hour and a half, and all of them maintained a similar structure. Each session began with a 15-minute meditation. This meditation was followed by a short discussion in which participants could explain their feelings and emotions during the previous meditation and the tasks they had completed during the week. Progress and difficulties during the week were shared, and then some theoretical aspects of how to relate to their thoughts, feelings, and emotions were explored. Finally, a new meditation based on the theme of the theoretical block was introduced. Finally, the homework for the following week was explained, which always included at least one daily meditation. Following the MBCT, instructions for each lesson were defined with a specific goal in order to train a different mental state. The titles and objectives of the eight sessions are given below: Session 1: Autopilot. The main objective is to introduce and explain the consequences of being on autopilot and the benefits of being in a state of mindfulness. Session 2: Facing the challenge. The main goal is to explore the first meditation experiences, giving the participants a metacognitive perspective. This working lesson is fundamental because challenges are faced, particularly all the obstacles that have appeared during their homework. Some of them would be “I could not find the moment to meditate”, “I get bored”, “I get really angry”, “I fell asleep”, “I am trying very hard, but I’m just not focusing enough”, etc. Session 3: Breathe awareness. The goal is to introduce the breath meditation as a link to the present moment when the mind is just wandering between thoughts, feelings, or emotions. Session 4: Remaining in the present. The goal is to observe reality, exploring new paths to relate to our own emotions, feelings, or thoughts. Session 5: Letting be. The goal is to develop a different relationship with the experiences without any kind of judgement. Session 6. Thoughts are not a fact. The goal is to encourage the participants to reduce their identification with their thoughts, learning to observe their thoughts just as events. Session 7. In what ways could I take better care of myself? The goal is to start closing the intervention; in this way, new strategies are sought in order to avoid relapse and learn techniques to take better care of ourselves. Session 8. Using what was learned. The goal is to learn how to maintain a practice after the intervention, introducing meditation as a lifestyle. Thus, the participants had learned a technique to learn how to face different low moods. Once the intervention is over, a summary of the intervention is given. On average, subclinical subjects who completed the intervention attended 97% of the scheduled sessions; average attendance in Groups 1 and 2 was 96.4% and 97.5%, respectively. Of the 23 participants who completed the intervention, 7 (30.4%) completed at least 9 (out of 10) sessions, and 15 (69.6%) completed all 10 sessions. The only participant who did not complete the intervention had an average attendance of 60% of the scheduled sessions. Subjects with moderate insomnia who completed the intervention attended 98.2% of the scheduled sessions; average attendance in groups 1 and 2 was 97.5% and 98.9%, respectively. Of the 25 participants who completed the intervention, 5 (20%) completed at least 9 (out of 10) sessions and 20 (80%) completed all 10 sessions. Table 3 Means and Simple Effects Tests for Significant Interactions between Subclinical Groups (control and intervention) at Pre-intervention and Postintervention  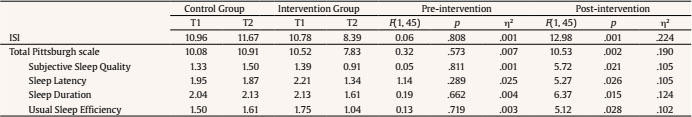 Analysis Mixed ANOVAS of 2 times (before versus after intervention; within subjects) x 2 groups (intervention versus control; between subjects) were performed to compare the baseline and post-intervention means. The data were analyzed with SPSS 21. Subclinical Group The main effects and interactions (time x group) in the subclinical group are presented in Table 2. Significant interaction effects were observed for the variables ISI, PSQI total, and the following dimensions: subjective sleep quality, sleep latency, sleep duration, and usual sleep efficiency. There were no significant interaction effects on the following PSQI dimensions: sleep disturbance, use of hypnotic medication, and daytime dysfunction (see Table 2). Simple effects tests were conducted to analyze the significant interactions in order to verify that there were no differences between the groups before starting the intervention and the possible differences between the groups at the end of the intervention. These simple effects tests confirmed that there were no between-group differences in any of the dependent variables at pre-intervention, but after the intervention significant differences between groups were found in all the dependent variables (see Table 3). Regarding the ISI, simple effects tests showed a significant decreasing effect in the intervention group, F(1, 45) = 10.41, p = .002, η2 = .188, whereas the effect was not significant in the control group, F(1, 45) = .95, p = .334, η2 = .021. Among the participants in the treatment group, based on the ISI cut-off point, 34.8% obtained a score equal to or lower than 7 on this scale at the end of the intervention and could be classified as not having significant insomnia. In the case of the PSQI, simple effects tests showed that the intervention group obtained significant decreasing effects on subjective sleep quality, F(1, 45) = 15.25, p = .027, η2 = .105; sleep latency, F(1, 45) = 25.71, p < .001, η2 = .364; sleep duration, F(1, 45) = 24.34, p < .001, η2 = .351; usual sleep efficiency, F(1, 45) = 8.23, p = .006, η2 = .115; and the PSQI total, F(1, 45) = 17.01, p < .001, η2 = .274, whereas the control group showed no significant effects on subjective sleep quality, F(1, 45) = .66, p = .419, η2 = .015; sleep latency, F(1, 45) = .24, p = .622, η2 = .005; sleep duration, F(1, 45) = .64, p = .425, η2 = .014; usual sleep efficiency, F(1, 45) = 1.68, p = .201, η2 = .036; or the PSQI total, F(1, 45) = 1.69, p = .199, η2 = .036. Moderate Group The main effects and interactions (time x group) in the moderate group are presented in Table 4. Significant interaction effects were observed for the following variables: ISI, PSQI total, and the following PSQI dimensions: subjective sleep quality, sleep latency, and sleep duration (see Table 4). There were no significant interaction effects on the following dimensions of the PSQI: usual sleep efficiency, sleep disturbance, use of hypnotic medication, and daytime dysfunction. Simple effects tests were conducted to analyze the significant interactions in order to verify that there were no differences between the groups before starting the intervention and possible differences between the groups at the end of the intervention. These simple effects tests confirmed that there were no between-group differences in any of the dependent variables at pre-intervention, but after the intervention significant differences between groups were found in all the dependent variables (see Table 5). Table 5 Means and Simple Effects Tests for Significant Interactions between Moderate Groups (control and intervention) at Pre-intervention and Post-intervention  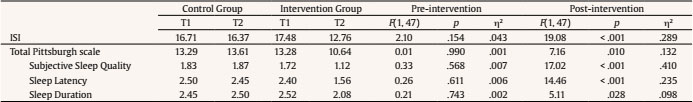 Regarding the ISI, simple effects tests showed a significant decreasing effect in the intervention group, F(1, 47) = 84.88, p < .001, η2 = .644. The effects were not significant in the control group, F(1, 47) = .41, p = .527, η2 = .009. Among the participants in the treatment group, based on the ISI cut-off point, 84% obtained a score between 8 and 14 on this scale at the end of the intervention and could be classified as having subclinical insomnia. Regarding the PSQI dimensions, simple effects tests showed that the intervention group obtained a significant decreasing effect on subjective sleep quality, F(1, 47) = 32.64, p < .001, η2 = ,410; sleep latency, F(1, 47) = 34.09, p < .001, η2 = .420; sleep duration, F(1, 47) = 15.05, p < .001, η2 = .243; and the PSQI total, F(1, 47) = 37.49, p < .001, η2 = .444. In the control group, the effects were not significant for subjective sleep quality, F(1, 47) = .15, p = .699, η2 = .003; sleep latency, F(1, 47) = .08, p = .788, η2 = .002; sleep duration, F(1, 47) = .13, p = .721, η2 = .003; or the PSQI total, F(1, 47) = .08, p = .778, η2 = .002. The present study aimed to evaluate whether an MBCT-based program for older adults with subclinical and moderate insomnia obtained significant improvements in sleep quality. The results provide evidence about the efficacy of this intervention in reducing the severity of insomnia symptoms assessed with the ISI scale and significantly improving the quality of sleep assessed with the PSQI. Regarding the insomnia severity, in the subclinical group, when comparing the groups, those who received the intervention significantly decreased their scores compared to their baseline; however, their symptoms were not reduced to below the clinical cutoff for insomnia disorder in all the participants. The comparison of the moderate insomnia groups confirmed that those who received the intervention showed a significant decrease in severity, emphasizing that their scores at the end were equivalent to those in the subclinical range. In this regard, and in relation to the effectiveness observed, the percentage of subjects in the moderate group who reduced their symptoms and progressed to the subclinical category was higher than the percentage of subclinical subjects who progressed to the category without significant insomnia. This observed difference in percentages indicates that this type of intervention appears to be more effective for this clinical group. In addition, effect sizes of the significant interactions in the moderate group were higher in the dimensions of subjective sleep quality and sleep latency, and a medium effect size was obtained even for the ISI scale, whereas in the subclinical group, the effect size was small. Some authors, such as Jiang et al. (2021), reported that even virtual mindfulness-based interventions can be as effective as evidence-based treatments and alleviate some aspects of moderate to severe sleep disorders. However, as Low et al. (2020) pointed out, intervening in subjects with mild symptoms is particularly important, given the large proportion of individuals in the community with subclinical insomnia. Moreover, in this case there is a greater likelihood that insomnia symptoms will progress to clinical levels if left untreated. Furthermore, the program improved the total sleep quality, measured by the PSQI. In the subclinical group, a significant improvement was achieved after completing the intervention in the following dimensions: subjective sleep quality, sleep latency, usual sleep efficiency, and especially sleep duration. In the case of moderate insomnia, the program was effective in the treatment group, improving the total score and the following dimensions: subjective sleep quality, sleep latency, and sleep duration. However, the results of the program did not present any significant improvements in sleep disturbances, use of hypnotic medication, or daytime dysfunction. Regarding the cognitive therapy administered to participants with insomnia in our intervention, it should be noted that there is ample evidence supporting the effectiveness of these techniques (e.g., CBT-I) in reducing scores on the sleep severity index (Arnedt et al., 2021; Cheung et al., 2019; Ho et al., 2015; McCurry et al., 2016; Zachariae et al., 2016) and improving sleep quality scores (Cheung et al., 2019). Furthermore, the meta-analysis by Selvanathan et al. (2021) also confirmed these results, which converge with those obtained in the present study. Specifically, Espie et al. (2007) and Sandlund et al. (2017) found significant improvements in sleep latency, a reduction in the number of awakenings after sleep onset, and improved sleep efficiency. Davidson et al. (2019) concluded that a sleep program with stimulus control and sleep restriction based on behavioral components showed better effects on sleep latency and awakenings after the onset of sleep, as well as on the general reduction in severity evaluated with the ISI scale. Cognitive components have also been shown to be effective in reducing insomnia by facilitating the identification and modification of dysfunctional thoughts, redirection of unrealistic expectations and misperceptions of sleep, elimination of worry, and modification of attentional bias (Harvey et al., 2014; Rosenberg et al., 2021). The CBT-I acts on the belief system and sleep-related maladaptive behaviors (Palagini et al., 2016). In addition, it reduces the factors that maintain long-term insomnia, such as behaviors that interfere with sleep, e.g., obsessive brooding (Carney et al., 2010) and cognitive attempts to control the sleep process and sleep-related anxiety (Baglioni et al., 2020). Regarding the use of mindfulness for insomnia, Kanen et al. (2015) stated that it mainly affects subjective aspects, such as some dimensions of the PSQI. These results were supported by Wang et al. (2020), who found significant improvements on the PSQI, except in the sleep efficacy dimension. According to our results, mindfulness practice significantly reduces the severity of insomnia measured by the ISI (Gross et al., 2011; Ong et al., 2014) The mindfulness technique for insomnia focuses on the positive or negative evaluation of thoughts, sleep-related beliefs, and selective awareness of sleep signs in order to be able to detect and accept them when the insomnia symptoms appear (Ong et al., 2012). Subjects with sleep disorders are characterized by high obsessive rumination, an internal and external bias about sleep problems, and negative thoughts, which lead to worse sleep quality (Harris et al., 2015). Mindfulness provides awareness abilities and attention (Tang et al., 2007), minimizing the difficulties. Thus, it can readjust cognitive processes that interfere with sleep, decreasing obsessive rumination, pre-sleep arousal, stress, and sleep latency (Kim et al., 2016; Lau et al., 2018; Winbush et al., 2007). Despite these theoretical proposals, there is no complete explanation for how mindfulness reduces insomnia problems (Wang et al., 2020). However, it has been recognized that the success of this technique is due to stress reduction and emotional regulation (Perez-Blasco et al., 2016; Zhang et al., 2015). In fact, Winbush et al. (2007) describe how the technique intervenes in cognitive processes that affect the sleep process. Getting subjects to focus on the present decreases arousal, thus reducing sleep symptoms. Finally, it should be noted that CBT-I and mindfulness converge as techniques in their results; however, there are differences between these two therapies. CBT-I could include some relaxation techniques to overcome anxiety. In this way, the patient would learn to relax, stopping the anxious thoughts that come to him/her. This should not be mistaken for meditation. When a person meditates, he or she is likely to enter a relaxed state, which, apart from being pleasant, is not the purpose of meditation. When someone is meditating they are not learning to stop their thoughts, but rather to deal with them. In addition, in relation to the differences, CBT-I implies changes that will eventually modify how the person feels. This would be the top-down path because it is necessary to make a cognitive and behavioral change prior to the emotional change. In Mindfulness, the path is the bottom-up. The patient begins to observe experiences that will modify his/her emotions, which will lead to behavioral and cognitive changes. Some studies have provided evidence that non-pharmacological treatments such as cognitive therapy and mindfulness combined are effective in treating insomnia. It is surprising that these kinds of techniques are not widely used (Qaseem et al., 2016). Thus, future investigations should focus on contributing results of this combined therapy (Ong et al., 2014) because, as some authors indicate (Vanhuffel et al., 2017; Zhao et al., 2020), it improves sleep quality. As a future line of research, it would be interesting to study whether each of these techniques affects insomnia factors in a different way. The results obtained seem to highlight that cognitive therapy would have a greater effect on sleep severity, whereas mindfulness would benefit subjective sleep quality. Furthermore, the importance of evaluating dimensions of mindfulness should be pointed out. Knowing which dimensions of mindfulness show effects after the intervention would indicate which ones are likely to have a positive effect on insomnia. In addition, given that this research showed significant effects in reducing insomnia in the clinical group, it would be interesting to test whether this efficacy is also observed in the severe insomnia group. As a main limitation, it should be noted that it would have been important to assess the possible depressive symptomatology of the participants. It has been suggested that depression and insomnia could be comorbid conditions. Clinical and epidemiological studies have shown that sleep disturbance is closely linked to major depression. However, they are not randomly associated; they are either causally related to each other and/or common causalities underlie the two disorders (Staner, 2010). In addition, the results of the systematic review by Gebara et al. (2018) concluded that insomnia treatments have an effect on reducing depression, with the effects being more pronounced in clinically depressed subjects. Therefore, it would have been interesting to test whether these effects were observed in this study. In conclusion, an MBCT-based program can improve sleep quality in subclinical and moderate insomniac older adults, diminishing insomnia symptoms and improving sleep quality. Moreover, the program implemented in the present study significantly reduces insomnia severity in moderate insomniac subjects, inducing improvements in the subclinical group as well, although to a lesser extent. That said, the importance of engaging patients with insomnia at sub-clinical levels, in intervention pathways, seems evident. Specifically, early treatment of sleep disorders through the MBCT-based programs in the present research could benefit a large part of the community, protecting them from more severe forms of insomnia. Conflict of Interest The authors of this article declare no conflict of interest. Cite this article as: Camino, M., Satorres, E., Delhom, I., Real, E., Abella, M., & Meléndez, J. C. (2022). Mindfulness-based cognitive therapy to improve sleep quality in older adults with insomnia. Psychosocial Intervention, 31(3), 159-167.https://doi.org/10.5093/pi2022a12 |
Cite this article as: Camino, M., Satorres, E., Delhom, I., Real, E., Abella, M., & Meléndez, J. C. (2022). Mindfulness-based Cognitive Therapy to Improve Sleep Quality in Older Adults with Insomnia. Psychosocial Intervention, 31(3), 159 - 167. https://doi.org/10.5093/pi2022a12
melendez@uv.es Correspondence: melendez@uv.es (J. C. Melendez).Copyright © 2026. Colegio Oficial de la Psicología de Madrid








 e-PUB
e-PUB CrossRef
CrossRef JATS
JATS






