
Abstinence Outcomes according to Age of a Blended Smoking Cessation Treatment with an App
[Los resultados de la abstinencia segĂşn la edad de un tratamiento combinado para dejar de fumar con una aplicaciĂłn mĂłvil]
Daniel Suárez-Castro1, Ana López-Durán1, 2, Carmela Martínez-Vispo1, 2, María Barroso-Hurtado1, Elisardo Becoña1, & 2
1Smoking and Addictive Disorders Unit, Faculty of Psychology, University of Santiago de Compostela, Spain; 2Institute of Psychology (IPsiUS), University of Santiago de Compostela, Spain
https://doi.org/10.5093/clh2026a8
Received 3 March 2025, Accepted 28 November 2025
Abstract
Background: Age is associated with smoking behavior. However, scarce research has explored its role in smoking cessation outcomes in digital interventions to quit. The present study aims to assess the effect of age on a blended smoking cessation cognitive-behavioral treatment that includes an App. Method: The sample (N = 154) was divided into two groups: 18-44 years old vs. 45 years or older, based on the mean age of the overall sample. Results: Participants aged 18 to 44 were more likely to be abstinent than those aged 45 years or older at the end of treatment and at the 6-month follow-up. There was a significant effect of age on point-prevalence abstinence over time (OR = 1.91). Conclusions: Findings suggest that age is a relevant variable in digital interventions to quit smoking and that including an App as an adjunct could be especially relevant for younger people seeking cessation treatment.
Resumen
Antecedentes: La edad se asocia con el hábito tabáquico. Sin embargo, pocas investigaciones han explorado su papel en los resultados de la cesación tabáquica mediante intervenciones digitales para dejar de fumar. El presente estudio tiene como objetivo evaluar el efecto de la edad en un tratamiento combinado cognitivo-conductual para dejar de fumar que incluye una aplicación. Método: La muestra (N = 154) se dividió en dos grupos: 18-44 años vs. 45 años o más, según la edad media de la muestra total. Resultados: Los participantes de 18 a 44 años tenían mayor probabilidad de ser abstemios que aquellos de 45 años o más al final del tratamiento y en el seguimiento de 6 meses. Se observó un efecto significativo de la edad en la prevalencia puntual de abstinencia a lo largo del tiempo (OR = 1.91). Conclusiones: Los resultados sugieren que la edad es una variable relevante en las intervenciones digitales para dejar de fumar y que la inclusión de una aplicación como complemento podría ser especialmente pertinente para las personas más jóvenes que buscan tratamiento para dejar de fumar.
Palabras clave
Abandono del hábito de fumar, Aplicación móvil, Diferencias de edad, Intervención combinadaKeywords
Smoking cessation, App, Age differences, Blended interventionCite this article as: Suárez-Castro, D., López-Durán, A., Martínez-Vispo, C., Barroso-Hurtado, M., & Becoña, E. (2026). Abstinence Outcomes according to Age of a Blended Smoking Cessation Treatment with an App. Clinical and Health, 37, Article e260722. https://doi.org/10.5093/clh2026a8
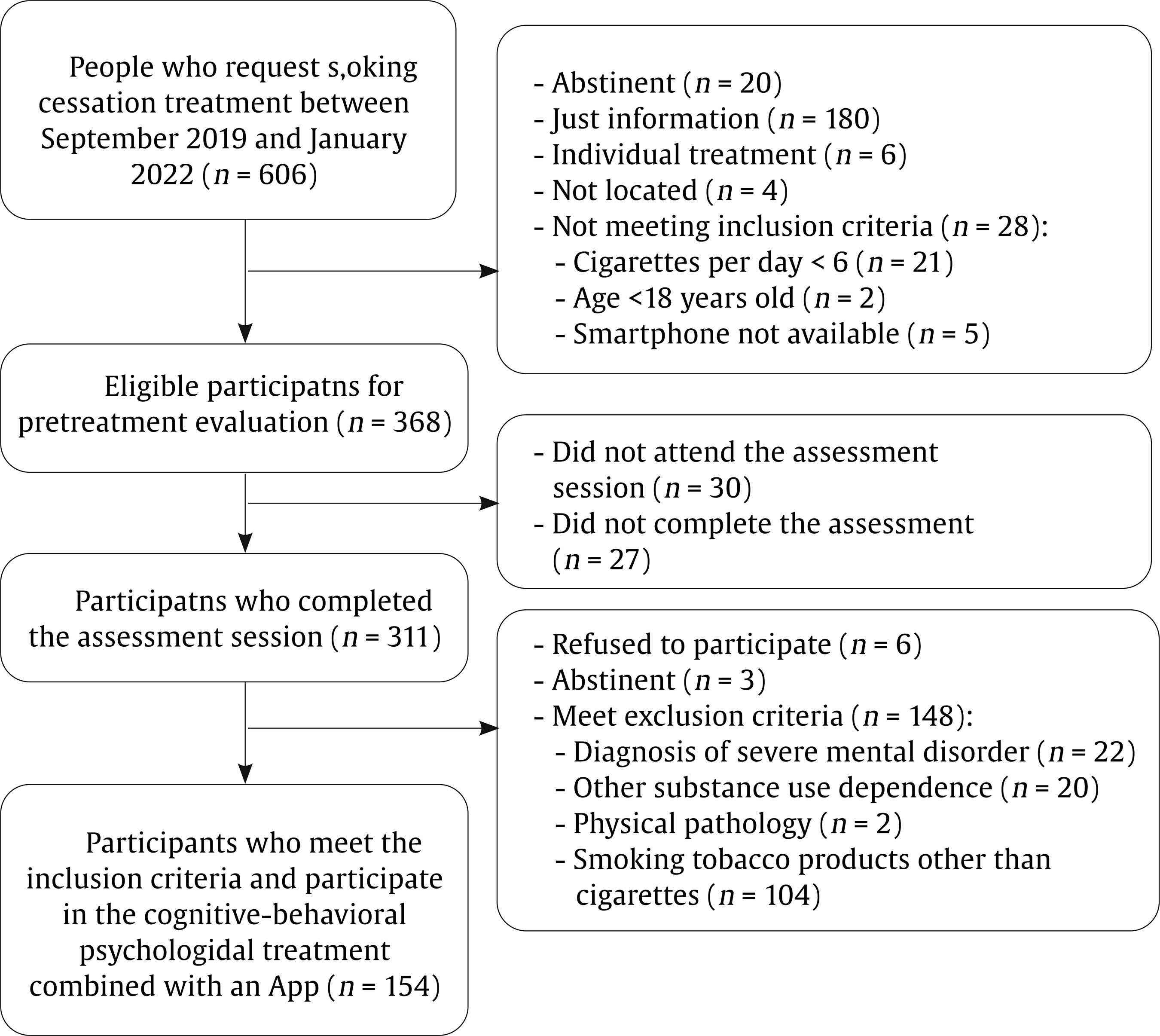
Correspondence: danielsuarez.castro@usc.es (D. Suárez-Castro).While smoking prevalence has decreased in recent years, many individuals still struggle with quitting smoking. Different variables have been analyzed for their relevance to cigarette use and the quitting process. One of these is sociodemographic variables, like smokers’ age, which has been studied from different perspectives. For instance, from an epidemiological perspective, age is relevant as prevalence rates tend to differ according to this variable. In this line, in Spain, the 25-34-years old age group has the highest prevalence of daily smoking (26.26%), followed by the 35-44 and 45-54 age groups, with prevalence rates of daily smoking of 24% approximately (Instituto Nacional de Estadística [INE], 2020). Regarding smoking behavior, young smokers tend to smoke fewer cigarettes per day and have lower levels of tobacco dependence than older smokers (Grant et al., 2020; Xu et al., 2022). In addition, younger smokers have lower rates of seeking smoking cessation treatment, which is particularly relevant because the age of cessation is the main predictor of the reversibility of tobacco-related harm (Fanshawe, 2017). Early smoking cessation improves overall health outcomes and reduces the risk of developing smoking-related diseases such as cancer or cardiovascular diseases. In this vein, Kenfield et al. (2008) showed that individuals who quit smoking before 35 have a 90% lower risk of developing coronary heart disease than those who continue smoking. Moreover, research has shown that the risk of mortality for individuals who quit smoking before the age of 40 is almost the same as that of a person who never smoked (Jha, 2020). Previous literature has indicated that younger smokers have specific motivations to continue smoking such as maintaining a positive social identity with other smokers, which could hinder quitting because smoking could facilitate social connections (Hoek et al., 2013). Previous studies have shown that younger smokers are less likely to enrol in smoking cessation treatments compared to those of an older age (Audrain-McGovern et al., 2007). This is consistent with national data from the Spanish National Health Survey, which highlights that smoking cessation treatment is more commonly sought by individuals aged 45 or older (INE, 2020). Differences according to smokers’ age have also been analyzed in relation to the outcomes of smoking cessation treatments. Age appears to play a significant role in treatment response, influencing both the likelihood of achieving and maintaining abstinence. Previous literature consistently highlights that older smokers often demonstrate higher rates of success in quitting compared to younger smokers (Ávila et al., 2022; Gallus et al., 2023). Several explanations have been proposed for this trend. For instance, older smokers may present higher levels of motivation to quit, smoking-related diseases, or receive physician recommendations which can reinforce adherence to cessation interventions (Sánchez et al., 2021). Therefore, smoking cessation programs targeting young adults—with components such as information related to that age, digital interactivity, and social identity reframing—are likely to be more attractive than generic approaches. Related to young people, smartphone App technology could be particularly attractive to younger smokers because Apps are widely used by this population (Ditrendia, 2022). Previous studies have focused on designing smoking cessation Apps by adapting conventional quitting interventions whose effectiveness has been previously established (Businelle et al., 2016; Hoeppner et al., 2021). Most existing studies have focused on the use of standalone self-help apps, showing mixed evidence since some findings suggest modest benefits, but long-term abstinence rates remain inconsistent (Barroso-Hurtado et al., 2021). However, there is limited research on incorporating apps as an adjunct of in-person smoking cessation treatment programs. Therefore, adding a smartphone App to a smoking cessation treatment could be a motivating and attractive feature to encourage young smokers to enroll in a smoking cessation program and quit smoking. Moreover, whereas prior research has explored self-help smoking cessation Apps, this study contributes to expanding the literature by evaluating a blended smoking cessation treatment underscoring the relevance of age as a significant sociodemographic factor. The aim of this study is to assess the influence of age on smoking cessation outcomes of a cognitive-behavioral smoking cessation treatment that uses an mHealth App as a complement. Setting This study was conducted at the Smoking Cessation and Addictive Disorders Unit of the University of Santiago de Compostela (Spain) between September 2019 and September 2022. The Bioethics Committee of the University of Santiago de Compostela approved the study (USC25-2020). This study is a secondary data analysis of two previously conducted studies (Barroso-Hurtado et al., 2024; López-Durán et al., 2025). López-Durán et al. (2025) was registered at clinicaltrials.gov (NCT04765813). Both trials followed a similar treatment protocol and procedures. Participants The overall participants’ flow chart is presented in Figure 1. To participate in the study, smokers had to meet a number of inclusion and exclusion criteria. Eligible participants were those who were at least 18 years of age, wished to participate in the smoking cessation treatment, had a smartphone, completed the pre-treatment assessment, smoked at least 6 cigarettes per day, and provided written informed consent. Participants were excluded if they reported having participated in similar treatment over the previous year or having received pharmacological smoking cessation treatment, other substance use dependence (e.g., cannabis dependence), smoking tobacco products other than cigarettes (e.g., e-cigarettes), the presence of a physical pathology that required immediate intervention (e.g., cancer), and/or a diagnosis of severe mental disorder (e.g., bipolar). Inclusion and exclusion criteria were assessed through self-report. Participants were recruited through the media, posters in healthcare centers, word of mouth, or they were referred to the unit by their primary care physician. Of a total 606 individuals who requested information to quit smoking, 154 met the abovementioned criteria and received the smoking cessation treatment at the Smoking Cessation and Addictive Disorders Unit of the University of Santiago de Compostela (Spain). Procedure Smokers interested in participating in the Smoking Cessation Program requested information by email, phone, or in person. An appointment was scheduled for the pre-treatment assessment session. During this session, a structured clinical interview was carried out, together with the completion of various questionnaires. Regarding the intervention, the cognitive-behavioral treatment Programa para Dejar de Fumar (A Quit Smoking program; Martínez-Vispo et al., 2019) combined with a smartphone App was carried out. Treatment elements were: self-report and graphic representation of consumption, nicotine fading, stimulus control, activities to prevent withdrawal syndrome, relapse prevention, and behavioral activation strategies. The smoking cessation program consists of eight weekly 60-minute treatment sessions in a group format. No individual sessions were conducted between weekly group sessions. All group sessions were conducted face-to-face. Sessions were performed in groups of 6-8 participants. Post-treatment assessments were carried out during Session 8 (end of treatment), and after these, face-to-face follow-ups were conducted at 3 and 6 months. The features of the App were: (1) self-report of cigarette consumption (each cigarette smoked is recorded, specifying the day, time, and situation) and (2) session material (written materials of the contents explained in each treatment session.) During the follow-up period, the features were: (1) gains and achievements (number of days abstinent, money saved, and time gained since quitting smoking), with the achievements section reporting health-related improvements to quitting; (2) tips to maintain abstinence: a sentence accompanied by a photo (e.g., do not have lighters nearby; do not feel guilty for having the urge to smoke); (3) SOS, resources to manage cravings including videogames, videos, gifs, and an emergency contact agency; and (4) my videos: recording videos to increase motivation to remain abstinent. Self-recordings of cigarettes and session materials were also included during the follow-up period. A more detailed description of both studies’ procedures can be found in Barroso-Hurtado et al. (2024) and López-Duran et al. (2025). Measures During the pre-treatment evaluation session, sociodemographic characteristics and smoking-related variables were collected through a face-to-face structured interview. We also applied the following instruments: Smoking Habit Questionnaire (Becoña, 1994) It consists of 59 items related to sociodemographic variables (age, sex, educational level, and marital status) and tobacco use. Specifically, this instrument collects data on cigarettes per day, tobacco brand, tobacco use history, maximum number of cigarettes smoked in a day, previous attempts to quit and illnesses caused by smoking, among other. Fagerström Test for Cigarette Dependence (FTC; Fagerstrom, 2012) It consists of 6 items for assessing cigarette dependence. The total score ranges from 0 to 10. Specifically, a score ≥ 6 indicates nicotine dependence. The Spanish version has a Cronbach alpha of .66. Customer Satisfaction Questionnaire-8 (CSQ-8; Vazquez et al., 2017) This is an eight-item self-report instrument that assesses overall satisfaction with the intervention program administered after the end of the intervention. One item was added to this questionnaire in to assess satisfaction with the mobile App. Satisfaction with the app was assessed both at the end of treatment and at follow-ups using the question with the maximum possible score being 3: “Overall, my level of satisfaction with the app is...” End of Treatment Questionnaire (Becoña & Míguez, 1995) This questionnaire was completed in the last treatment session. The following aspects are assessed: brand and number of cigarettes smoked that day and the previous day (if the person is still smoking); planned quit date (if they are still smoking); physical and psychological improvements experienced after the treatment; and a self-assessed confidence rating in remaining abstinent from smoking over the next six months. Follow-up Questionnaire (Becoña & Míguez, 1995) This questionnaire was completed at the 3- and 6-month follow-ups. This questionnaire has two versions: a) one for those participants who are abstinent and that assess whether they have smoked any puff or cigarette since the end of treatment, how long they have been abstinent and the date they smoked their last cigarette) and b) other version for those participants who are smoking, assessing the number of cigarettes they smoke per day and tobacco brand, whether they have made other quit attempts; duration of those quit attempts, among other). Abstinence Participants were considered abstinent if they reported abstinence, not even a puff of combustible tobacco products (i.e., cigars, rolled cigarettes) or other nicotine-related products (i.e., e-cigarettes) for ≥ 7 days at the end of treatment and at the 3-month follow-up and for ≥ 30 days at the 6-month follow-up. Participants were considered smokers if they reported smoking during the follow-up sessions or if they did not were reached. Because of the COVID-19 pandemic, abstinence was only biochemically validated through carbon monoxide (CO) for 17.50% of the participants. Table 1 Baseline Sociodemographic and Tobacco-related Variables according to Age  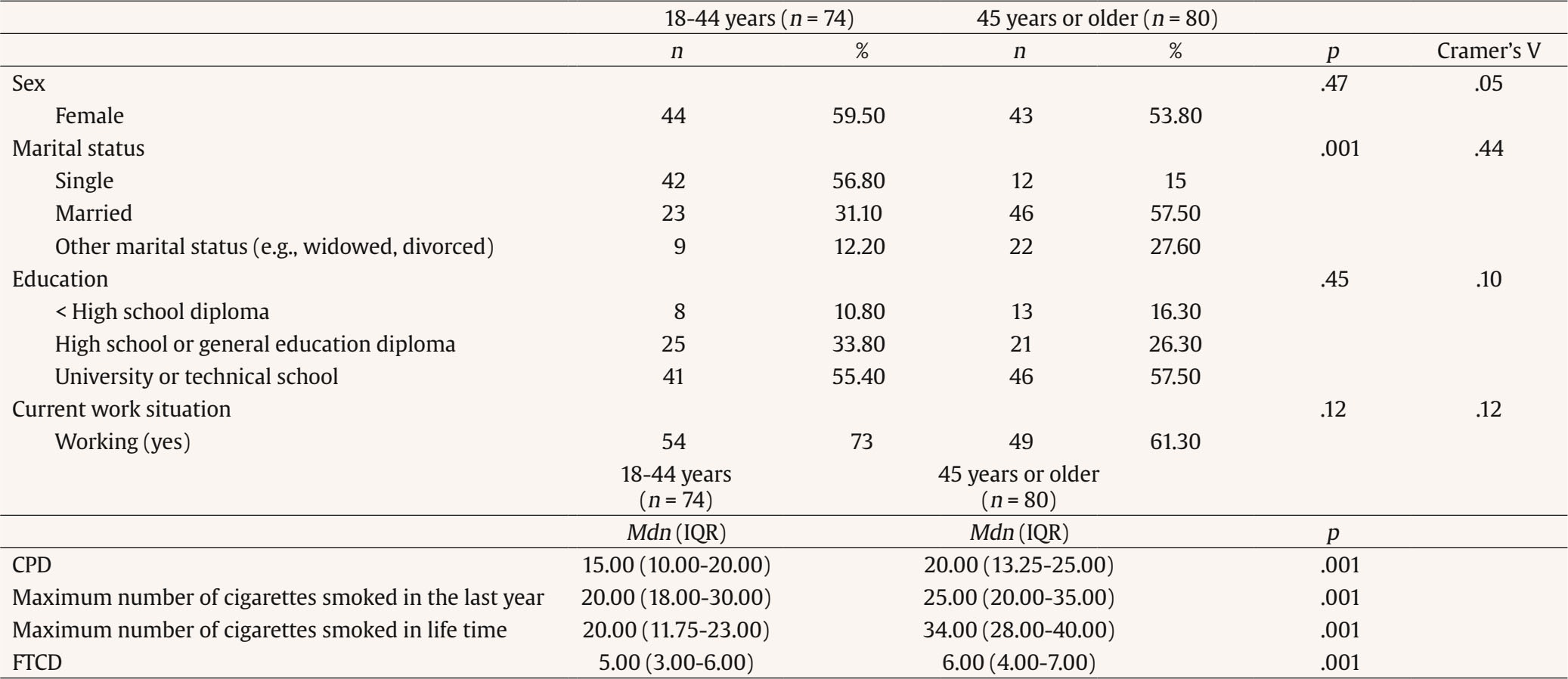 Note. CPD = cigarettes smoked per day; FTCD = Fagerström Test for Cigarette Dependence. Statistical Analysis Descriptive statistics of sociodemographic and smoking-related characteristics are reported as means with standard deviations or frequencies with corresponding percentages. For data analysis, the sample was divided into two age groups (young + early middle age vs. late middle age + older adults). This grouping was done to ensure sufficient statistical power and to create more balanced group sizes, as the percentages of participants in the youngest and oldest original categories were very low. The source of the data was statistically controlled for in all analyses. Student’s t-test was used to examine the differences according to age (18-44 years vs. 45 years or more) in continuous variables such as tobacco variables and number of cigarettes smoked daily at the end of the treatment and at the 3- and 6-month follow-ups. To determine effect sizes, we used Cohen’s d. The Mann-Whitney U statistical test was used when data were non-normally distributed. Chi-square tests were used to evaluate differences between the age groups (18-44 years vs. 45 years or more) in categorical variables such as sociodemographic and tobacco variables, abstinence rates at the end of treatment and at the 3- and 6-month follow-ups, and treatment adherence. Cramer’s V was used to determine effect sizes. Generalized Estimating Equations (GEEs) were used to assess the association of participants’ age with abstinence over time. This analysis allows handling correlated data, such as repeated measurements and provide robust standard error. GEE models, which avoid assumptions about random effects and apply robust variance estimates, help minimize Type I error across timepoints (Lee et al., 2007). We performed an unadjusted model and an adjusted model by studies (Barroso-Hurtado et al., 2024; López-Durán et al., 2025), number of cigarettes smoked per day, cigarette dependence, sex and marital status (significant differences variables at baseline). The analysis focused on evaluating the main effects of age (18-44 years old vs. 45 years or older) and time points (end-of-treatment, 3- and 6-month follow-ups) on the prediction of abstinence rates (0 = smoker and 1 = non-smoker). The model was fitted using an unstructured working correlation. Statistical analyses were performed using the SPSS version 27 for Windows. The value of the significance level was set at .05. Age Differences: Descriptive Analysis Total sample (N = 154) participants’ mean age was 45.03 years (SD = 10.54), with 56.50% females. Regarding marital status, 44.80% were married or living with a partner. Most participants had higher education (56.50%) and 66.90% were working at the time of pre-treatment assessment. Baseline sociodemographic and tobacco-related variables according to age groups are shown in Table 1. The mean age of the 18-44-year-old group was 36.28 (SD = 6.37), whereas the mean age of the 45 years or older group was 53.13 (SD = 6.31). Significant differences were found between age groups in marital status (χ2 = 1.59, p = .001; Cramer’s V = .44), cigarettes smoked per day (U = 2103, p = .001), maximum number of cigarettes smoked in the last year (U = 2203, p = .001), maximum number of cigarettes smoked in lifetime (U = 2173.5, p = .001), and Fagerström Test for Cigarette Dependence (FTCD) (U = 2118, p = .001). Adherence to the treatment was high, 78% of the participants completed all sessions. 58 participants from the young/middle age group and 62 participants of the older age group. Regarding follow-ups, 17.6% of participants aged 18-44 did not attend the 3-month and 6-month assessments. In the ≥ 45-year-old group, 25% missed the 3-month follow-up and 23.8% missed the 6-month follow-up. No significant differences were found between the two age groups (χ² = 2.57, p = .27). Smoking Cessation Rates according to Age At the end of treatment, 66.20% of the participants were abstinent, 37% at the 3-month follow-up, and 33.80% at the 6-month follow-up. Abstinence rates for each age group are shown in Figure 2. Significant differences between age groups were found at the end of treatment (χ2 = 4.17, p = .04, Cramer’s V = .16) and at the 6-month follow-up (χ2 = 4.21, p = .04, Cramer’s V = .16). No significant differences were found at the 3-month follow-up (χ2 = 3.51, p = .06). Figure 2 Differences between Adults Aged 18–44 vs 45 or Older in 7-day Abstinence Rates at End-of-treatment, 3-month Follow-up, and 30-day Abstinence Rates at 6-month Follow-up.  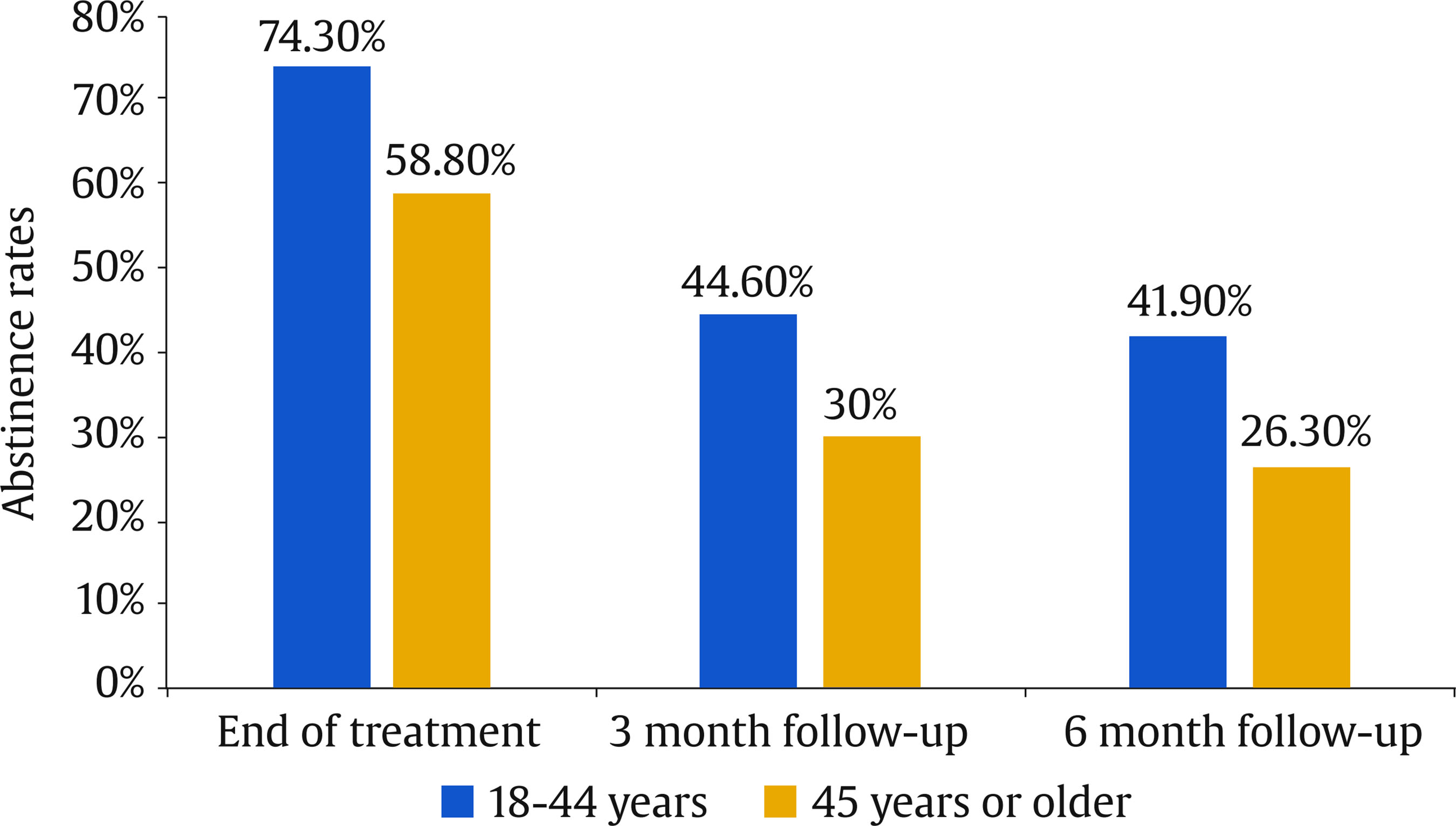 Influence of Age on Smoking Cessation The GEE modeling of the main effects of age and time showed a significant effect of age and time on point-prevalence abstinence in the unadjusted and adjusted model (Tables 2 and 3). The age group of 18-44 years was more likely to be abstinent over time than the age group of 45 years or older. In addition, time was significant, meaning that the probability of abstinence decreased across follow-ups. Table 2 Generalized Estimation Equations (GEE) predicting point-prevalence smoking abstinence   Note. CI = confidence interval; OR = odds ratio. *p < .05, **p < .01. Table 3 Generalized Estimation Equations (GEE) Predicting Point-prevalence Smoking Abstinence Adjusted by Marital Status  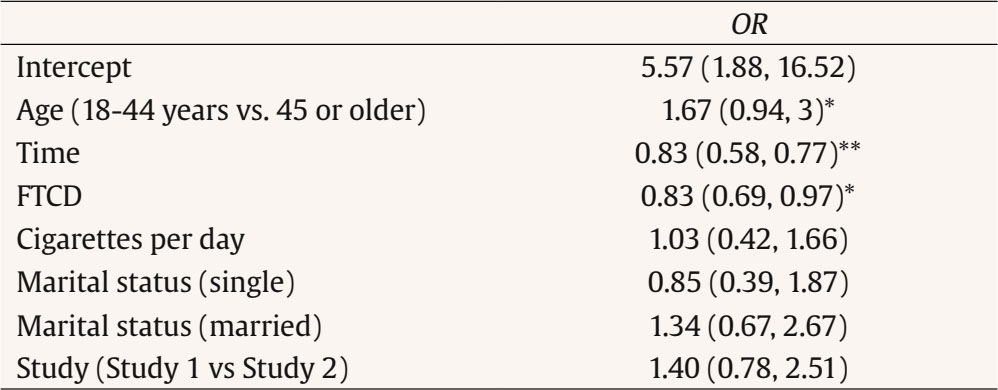 Note. CI = confidence interval; OR = odds ratio; FTCD = Fagerström Test for Cigarette Dependence; comparison group to marital status: other marital status (e.g., widowed, divorced) *p < .05, **p < .01. Satisfaction with the Blended Intervention and with the App Satisfaction with the treatment was high, with a mean score of 31 (SD = 1.82) out of a possible 32 points. Among the 120 participants who completed the treatment, 73.40% rated the service received as excellent, 73.40% stated they would repeat the program if needed, and 72.10% indicated they would recommend the program to a friend. Significant age group differences were found participants aged 18-44 years reported higher overall satisfaction with the treatment compared to those aged 45 years or older (t = 2.5, p = .01, Cohen’s d = .45) Mean scores for overall satisfaction were above 2 at all measurement points, with the maximum possible score being 3. No differences were found between age groups (t = .62, p = .53) The aim of this study was to assess the effect of age on smoking cessation outcomes of a cognitive-behavioral smoking cessation treatment using an App as an adjunct. Overall, percentages of cessation success in the present study align with previous research that use Apps as an adjunct to smoking cessation treatment (Asayut et al., 2022; O’Connor et al., 2020). When examining the role of age, our findings showed that abstinence rates were significantly higher in younger smokers than in older smokers at the end of treatment and at the 6-month follow-up. While the difference between age groups was non-significant (p = .06), this may be due to the sample size, rather than a substantial shift in the underlying effect. Moreover, age was a significant predictor of point-prevalence abstinence across time, with younger age being associated with a greater likelihood of quitting smoking. These findings contrast with previous literature where older smokers often show greater cessation success (Ávila et al., 2022; Gallus et al., 2023). A possible explanation for this discrepancy may be the nature of the intervention. Specifically, we included a smartphone App as an adjunct to the cognitive-behavioral smoking cessation intervention, which could have enhanced engagement and motivation among younger participants as this age group tend to be more familiar and comfortable with digital tools, such as mobile Apps (Ditrendia, 2022). In fact, previous studies have shown that mHealth Apps are of particular interest to young people (Wartella et al., 2016), who often use Information and Communications Technology (ICT) to seek health-related information (Lupton, 2021). Moreover, research on digital interventions to quit smoking has shown promising outcomes for smoking cessation Apps, indicating that younger adults tend to show greater engagement and success compared to older groups (Baskerville et al., 2018; Zhou et al., 2023). Therefore, these tools may align with younger users’ preferences, contributing to higher treatment adherence and abstinence rates in this group. Further research is needed to establish the impact of age on cessation outcomes in digital interventions, as the absence of a control condition without a mobile App prevents us from isolating its specific contribution to smoking cessation outcomes by age. Moreover, other variables should be considered when interpreting these outcomes. Significant differences between age groups were found in smoking behavior variables assessed at pre-treatment, such as cigarettes smoked per day and cigarette dependence levels, which align with previous studies indicating that younger smokers smoke fewer cigarettes per day and report lower cigarette dependence than older smokers (Grant et al., 2020; Xu et al., 2022). These differences at baseline could be explained by the fact that smoking for more years could produce a desensitization of nicotine receptors, potentially increasing dependence and cigarette use over time (Han et al., 2023). Since heavier smoking and greater dependence are both associated with reduced quit success (Doran et al., 2023), we run the models controlling for these variables, and our results remained consistent. This finding suggests that age-related differences in cessation outcomes are not solely explained by cigarette use intensity or dependence. More research is needed to determine whether quitting motivation and intervention engagement could be improved among younger participants due to the App inclusion or whether other variables, such as social support or health status are influencing these results. There are several limitations to be considered in this study. Firstly, the sample was recruited from September 2019 until January 2022. In this period, there were restrictions as a consequence of the COVID-19 pandemic and, therefore, it was not possible to obtain biochemical validation of abstinence for the entire sample. This was only obtained for 17.50% of the participants. Despite this limitation, to gain confidence in self-reported abstinence rates, we compared the abstinence rates at the end of treatment and the participants’ follow-ups with and without biochemical verification (only self-reported), and no significant group differences were found. Secondly, the sample comprises smokers seeking treatment for smoking cessation, so the outcomes obtained cannot be generalized to the general population of people who smoke. Thirdly, the sample size is limited and studies that replicate these results with larger samples are needed in order to provide more accurate results. Finally, establishing the cutoff age at 45 years old, despite that being based on the mean age of the total sample and previous studies conducted in Spain (López-Núñez et al., 2016; Martínez-Vispo et al., 2019), may introduce some biases and limit the ability to compare our data with previous studies using different age cutoffs and to capture more specific age-related effects. Despite the limitations, this study has some strengths. First, it adds information about the influence of the age of participants who seek smoking cessation treatment on abstinence achievement and maintenance. Despite the influence of age when attempting to quit smoking, few studies show abstinence outcomes according to participant’s age. Second, this study provides the outcomes of a blended treatment to quit smoking, combining a smoking cessation treatment with an App as a complement. As the development of Apps for smoking cessation is very recent (Barroso-Hurtado et al., 2021; Zhang et al., 2023), information about abstinence rates of this kind of treatment is relevant. Third, this study was conducted with a large clinical sample of smokers, expanding the previous literature about blended smoking cessation treatment with Apps that uses smaller sample sizes (Dan et al., 2016; Hertzberg et al., 2013). Finally, the abstinence rates obtained (66.20% at the end of treatment, 37% at 3 months, and 33.80% at 6 months) are high and align with previous research that showed abstinence rates of behavioral treatments to quit smoking (Patnode et al., 2021). In summary, the findings of this study have important clinical implications, contributing to understanding the factors that may influence the outcomes of smoking cessation treatments and obtaining higher abstinent rates for young smokers compared to older smokers. Including an App as an adjunct to a smoking cessation treatment may be a strategy to motivate younger smokers to quit smoking. More studies are needed to draw conclusions on how digital technologies may influence young people’s enrolment in smoking cessation treatments and their cessation outcomes. In addition, examining engagement-related variables—such as time spent on specific app features or frequency of use—could provide valuable insights into the app’s contribution to treatment effectiveness. This information would help in designing smoking cessation programs tailored to the needs of different age groups. Furthermore, investigating aspects such as digital literacy and patterns of interaction with app features could support the development of more personalized interventions for people who smoke.
Conflict of Interest The authors of this article declare no conflict of interest. Cite this article as: Suárez-Castro, D., López-Durán, A., Martínez-Vispo, C., Barroso-Hurtado, M., & Becoña, E. (2026). Abstinence outcomes according to age of a blended smoking cessation treatment with an app. Clinical and Health, 37, Article e260722. https://doi.org/10.5093/clh2026a8 Funding This work was supported by a predoctoral grant at the first author of this paper [grant number ED481A-2019/084] and by a grant of research at the other authors of the paper [grant number grant number [ED431B-2022/024], both by the Consellería de Cultura, Educación e Universidades of Xunta de Galicia (Spain). The data underlying this article will be shared on reasonable request to the corresponding author. References |
Cite this article as: Suárez-Castro, D., López-Durán, A., Martínez-Vispo, C., Barroso-Hurtado, M., & Becoña, E. (2026). Abstinence Outcomes according to Age of a Blended Smoking Cessation Treatment with an App. Clinical and Health, 37, Article e260722. https://doi.org/10.5093/clh2026a8
Correspondence: danielsuarez.castro@usc.es (D. Suárez-Castro).Copyright © 2026. Colegio Oficial de la Psicología de Madrid








 e-PUB
e-PUB CrossRef
CrossRef JATS
JATS







